The single-window shop for PCB Manufacturing and PCB Assembly In USA
PCB Trace Technologies Inc. is your single-window shop for any type of Printed Circuit Board Manufacturing and Full Turnkey PCB Assemblies in USA. Our expertise and specialization are in Engineering Prototypes, high layer count PCBs, and the entire extent of Electronics Manufacturing Services. We build every PCB to the top-quality standards. This includes Rigid PCBs, Flex PCBs, Rigid-Flex PCBs, and HDI PCBs. We build All PCBs and assemblies complying with ISO 9001:2015, ISO 13485:2016, J-STD-001, IPC-A-610E Our Electronic Assembly Services are unmatched in quality, speed, and workmanship. From bare circuit boards to final assemblies to box builds, PCB Trace Technologies Inc. is your premier single-window shop. We offer the most competitive pricing in the industry with a complete commitment to total customer satisfaction.
Contact us today for any type of PCB requirement for your project. As an eminent PCB manufacturer, PCB Trace offers a full gamut of services.
PCB Design Services
We offer a complete design service, starting from schematic capture, Bill of Materials generation, and printed circuit board layout.
Depending on your application, we will help you with PCB material selection so that your board performs to the optimum level. Whether your application requires high-frequency materials, low-frequency materials, high Tg, or low Dk, we will select the most effective material that will give you the best performance.
PCB Prototype Services
At PCB Trace, we understand your requirement of prototypes before you commit to full-scale manufacturing quantities. We will provide you with prototypes so that your design team can prove their concept in reality. We will also provide you with a free DFA/DFM report so that your design team can optimize their design to achieve a smooth production session.
PCB Fabrication Services
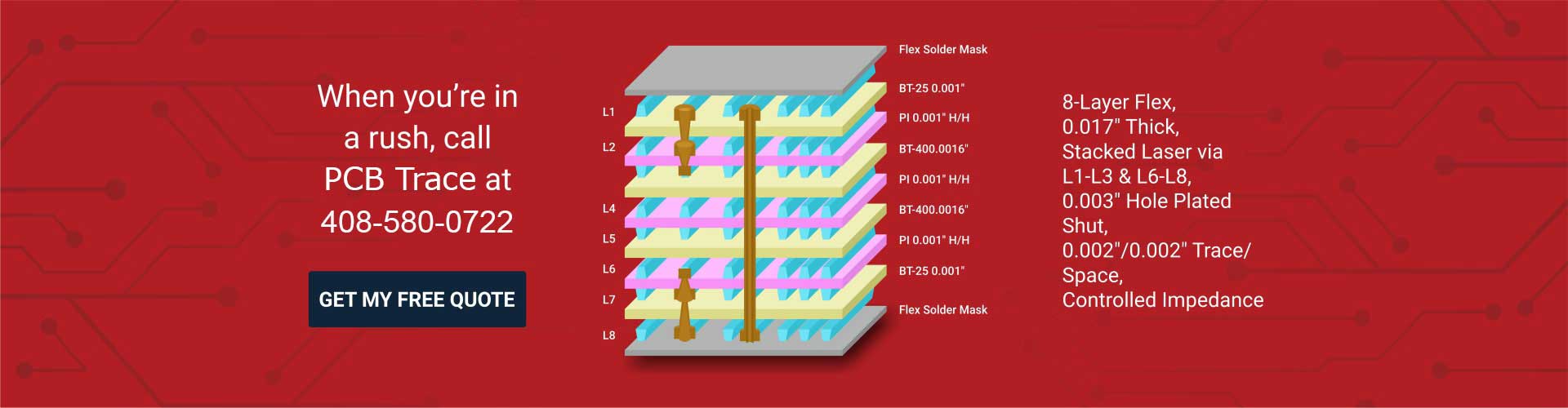
Our state-of-the-art PCB fabrication machinery offers you the highest quality and reliable printed circuit boards. We optimize your boards for signal integrity for high-frequency coverage or heavy copper for high current requirements. Whether your requirement is for single-sided, double-sided, multi-layered, or HDI PCBs, we have you covered.
PCB Assembly Services
As a part of printed circuit board complete solutions production unit in the USA, PCB Trace also offers the latest PCB assembly services for the board we have manufactured for you. We offer partial or full turnkey assembly services.
Under partial turnkey services, we accept the components you offer and purchase the balance to begin the assembly.
Under full turnkey services, we purchase all the components listed in your BOM and begin the assembly.
We offer assembly services for all components, whether Through Hole Technology (THT) or Surface Mount Technology (THT). For soldering THT components, we use a solder bath, and for SMT components we use a reflow machine. We can use leaded or lead-free solder as your application demands.
As a highly effective PCB supplier, you will find our PCB assembly costs the most affordable on the market. This is because we use the latest technology for assembling and soldering your boards.



















 Watch Video
Watch Video